States of matter are the different conditions, both physically and chemically, in which matter can exist. The states of matter are defined based on the kinetic energy and other important properties of the constituent particles within it. For example, in a gas, the molecules are distantly spaced and have very high kinetic energy. This leads the state to exist in a condition of increased volume. The thermodynamically defined states of matter are;
-
SOLID
- The constituent molecules have the lowest kinetic and stick together in minimal volume.
- Examples include ice, stones, etc.
-
LIQUID
- The constituent molecules have higher kinetic energy compared to solids but can flow freely due to expansion in volume.
- Examples include water, blood, etc.
-
GAS
- The constituent molecules have the highest kinetic energy compared to the other states, hence causing the individual molecules to stay far away from each other, increasing fluidity and volume.
- Examples include steam, Helium, etc.
It is possible to alter the kinetic energies of the molecules by various processes, and one of the simplest is by changing the temperature. This process would include either heating a substance to a higher temperature to increase the kinetic energy of the molecules or cooling a substance to a lower temperature to decrease the kinetic energy.
One such process is sublimation. The definition of sublimation states that the thermodynamic process of converting a solid to a gas directly without a liquid intermediate being formed is called so. We do not observe sublimation in most substances, and it is an exception out of the usual processes of inter conversion of the states of matter.
When we keep naphthalene balls between clothes to preserve them, and they evaporate without leaving any liquid residue, it is an example of sublimation.
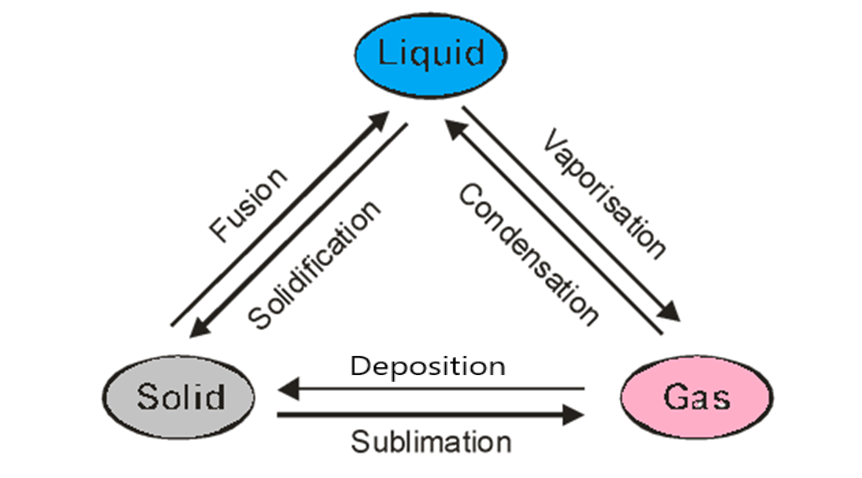
Sublimation Meaning?
Sublimation is the transition of a substance directly from the solid to the gas state without passing through the liquid state. The process of sublimation is a thermodynamically endothermic process that occurs at temperatures and pressures below the lowest pressure at which the substance can exist as a liquid, referred to as its triple point. This means that it absorbs heat to complete the process. The reverse process of sublimation is deposition or de-sublimation, in which a substance passes directly from a gas to a solid phase. Sublimation is also used as a generic term to describe a solid-to-gas transition (sublimation) followed by a gas-to-solid transition (deposition).
In simple words, sublimation is the process during which a solid is heated and gets converted to gas directly without producing the liquid intermediate. As mentioned in the earlier paragraphs, during sublimation, the molecules absorb energy from the heat supplied to raise the temperature. Henceforth causing the kinetic energy to increase for the individual molecules and make them rush farther from each other. This, in turn, will increase the volume sufficiently and convert the compact solid into a fluid gas.
What Is Endothermic Or Exothermic Process?
Thermodynamic processes are the ones that usually involve a change or flow in heat between substances. These processes are classified into two types. They are as follows;
- ENDOTHERMIC
- These processes include absorption of heat or energy in the system. The ‘endo’ translates to ‘inside,’ so the substance absorbs heat. This will cause the temperature to increase.
- Examples include boiling water to produce steam, etc.
- EXOTHERMIC
- These processes include the release of heat or energy in the system. The ‘exo’ translates to ‘outside,’ so the substance releases heat. This will cause the temperature to decrease.
- Examples include freezing of water to produce ice, etc.
There are intermediate processes too. The image below shows the differences between an endothermic and exothermic process.
/endothermic-and-exothermic-reactions-602105_final-c4fdc462eb654ed09b542da86fd447e2.png?resize=896%2C672&ssl=1)
Is Sublimation Endothermic Or Exothermic?
Sublimation is an endothermic process since it requires some form of energy to convert the solid into a gas component. Heat energy is usually supplied to bring about the physical change from a solid to gas during the process of sublimation. In other words, sublimation occurs due to the absorption of heat. This provides sufficient energy for the solid molecules to break the intermolecular attraction of their particles. Henceforth transitioning into the vapor phase.
In simpler words, to increase the kinetic energy of the constituent molecules to cause sublimation, we supply the energy from outside to raise the energy of the molecules and make them rush apart, increasing the volume. Henceforth, this would lead to the conversion of the state, and this will convert the solid to a gas.
Read Also: Chemiosmosis
The absorption of heat provides enough energy for some molecules to overcome the attractive forces of their neighbors and escape into the vapor phase, resulting in sublimation. It’s an endothermic change because it necessitates more energy.
In other words, sublimation occurs as a result of heat absorption, which provides enough energy for the solid molecules to break the intermolecular attraction of their particles and transition into the vapor phase. Thus, it is an endothermic process.

Sublimation Examples
The following are the examples of sublimation;
- The best example of sublimation is dry ice which is a frozen form of carbon dioxide. When dry ice gets exposed to air, dry ice directly changes its phase from the solid state to the gaseous state, which is visible as fog. Frozen carbon dioxide in its gaseous state is more stable than in its solid state.
- Another well-known example of sublimation is naphthalene which is an organic compound. Pesticides such as mothballs contain naphthalene in them. This naphthalene creates a toxic environment for the insects and kills them. At a temperature of 176F naphthalene sublimes to form vapors. It de-sublimates at cool surfaces to form needle-like crystals.
- Sulfur undergoes sublimation into poisonous and suffocating gases between 25°C and 50°C.
- Room fresheners that are found in toilets undergo sublimation.
- Iodine undergoes sublimation from a solid to a poisonous purple gas at 100 °C.
And many others.
Sublimation Applications
- Camphor is another substance that sublimes to give a very pleasant smell. Printing presses have specialized printers that work on the process of sublimation. In the process of printing, the ink turns from solid to gas and then again into the solid phase. The textile industry also uses this technique for the job of fabric printing.

- Freeze-drying is the sublimation process used in the frozen food industry to dehydrate the food. The frozen water in the material will sublimate from the solid phase to the gas phase when you reduce the surrounding pressure. We don’t need to add heat to remove water. Sublimation results in a high-quality product because it makes use of extremely low temperatures. The shape of the product remains in its original form, and once we rehydrate the product, it has excellent quality. Foodstuffs can be preserved for a longer time using the freeze-drying technique. NASA scientists use this method to preserve food for a longer time for astronauts.
- Dye-sublimation printers are now replacing inkjet printers. The dye sublimation process prints synthetic fabrics like polyester in the textile industry.